From lab to market: IISc team, surgeon develop dissolving stent to cut 2nd surgeries | India News

BENGALURU: A small, dissolvable tube developed in a Bengaluru lab is moving a step closer to hospital use, offering a way to avoid repeat procedures after major abdominal surgery.The device, called the “Asthana Stent”, is the result of a collaboration between researchers led by liver transplant surgeon Sonal Asthana of Aster CMI Hospital and Kaushik Chatterjee of IISc, spanning its Department of Materials Engineering and Bioengineering.It has now been licensed to Advanced Medtech Solutions Pvt Ltd under an Indian patent, moving from a laboratory prototype to a product that could reach the operating room.The idea originated from a recurring problem seen after liver transplants. Surgeons must join the bile duct, a delicate connection that can later leak or narrow. These complications affect 11% to 40% of patients and often require another procedure to correct.
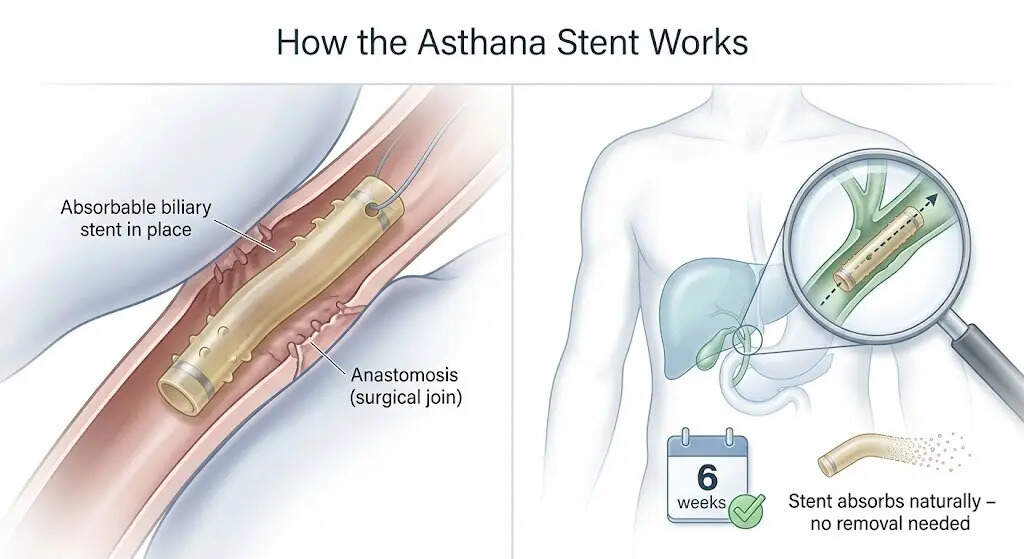
Existing solutions rely on plastic tubes, or stents, to keep the duct open while it heals. But this comes with trade-offs. Some extend beyond the body and require careful management. Others remain inside but must be removed later through an additional procedure, adding cost and risk.“The new stent takes a different approach. It is made from polydioxanone (PDS), a biodegradable polymer, which is already used in dissolvable surgical sutures. Once placed inside the body, it stays open in the duct for about six weeks, then slowly disintegrates and is absorbed. No removal is required,” the research team said.The engineering work of Thaseeb Rehman of the Department of Materials Engineering and Saswat Chaudhary of the Department of Bioengineering at IISc is needed to turn this concept into a functional device. The team designed the tube to stay open under pressure and prevent migration using surface ridges and Velcro-like hooks, while a flexible mid-section allows for placement even in the wrong-number of duct openings. Tiny radiopaque markers allow doctors to track it using X-rays.“Tests have shown that the stent can withstand pressures of more than 16 newtons, which is more than the bile ducts experience. It retains its structure for six weeks in both lab conditions and human bile, covering the critical healing window,” the researchers said.With the technology now licensed, the focus shifts to manufacturing and approval. The firm will undergo regulatory checks prescribed by the Central Drugs Standard Control Organization (CDSCO). The project also received support from the Indian Council of Medical Research (ICMR).If approved, the stent will be used during surgery and is available in multiple sizes. For patients, the benefit is straightforward: one operation instead of two and less chance of complications after going home.






